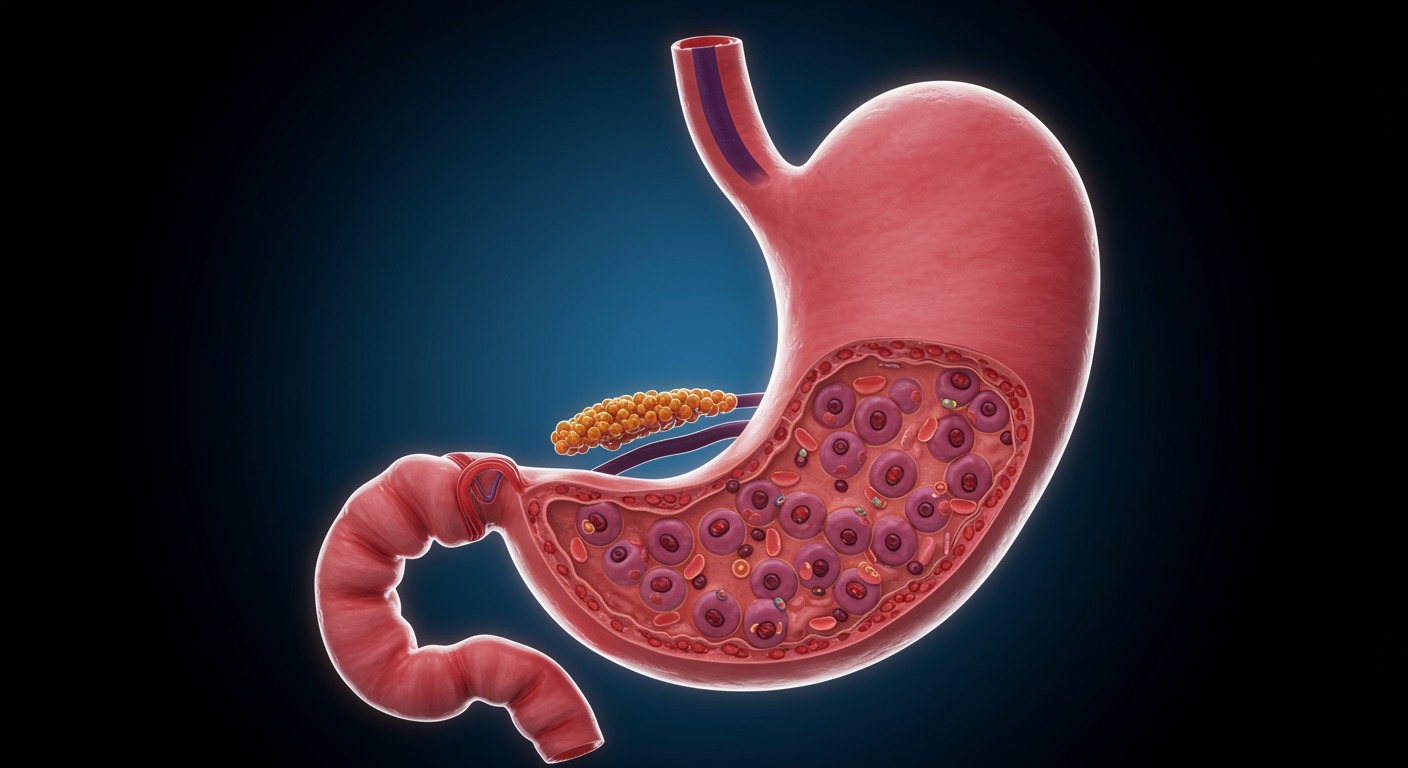
Every time you eat a steak or a bowl of lentils, your stomach launches a powerful chemical process. The role of HCl in stomach during protein digestion is absolutely central — without hydrochloric acid, your body simply cannot break down proteins into usable amino acids.
Contents
- 1 What Does Hydrochloric Acid Do to Proteins in the Stomach?
- 2 Does HCl Help Digest Protein Directly?
- 3 How Does Acid Kill Bacteria?
- 4 What Happens If There Is Too Much — or Too Little — Acid?
- 5 Age and HCl: A Factor People Overlook
- 6 FAQ
- 7 What Are the 3 Main Functions of HCl in the Stomach?
- 8 Does Animal Protein React Differently to HCl Than Plant Protein?
- 9 How Does Acid Protect the Stomach Lining?
- 10 Final Thoughts
What Does Hydrochloric Acid Do to Proteins in the Stomach?
HCl is secreted by parietal cells in the stomach lining, producing roughly 2 liters of gastric juice daily. It creates an intensly acidic environment with a pH of 1.5–3.5.
At the molecular level, HCl disrupts hydrogen bonds, ionic interactions, and disulfide bridges that hold a protein’s 3D structure together. This process — called denaturation — unfolds tightly coiled proteins, exposing peptide bonds hidden inside. Without this step, digestive enzymes can’t access the bonds they need to cut.
Does HCl Help Digest Protein Directly?
Not exactly. HCl itself doesn’t cleave peptide bonds. Its real job is activating pepsinogen — an inactive enzyme released by chief cells — into pepsin. Pepsin works best at pH 1.5–2.0 (slightly lower than the general stomach pH), and it chops denatured proteins into smaller peptide fragments.
So the chain looks like this: food enters stomach → HCl denatures protein → HCl converts pepsinogen to pepsin → pepsin breaks peptides → mixture moves to small intestine → pancreatic proteases (trypsin, chymotrypsin) finish the job → free amino acids absorb into the bloodstream.
How Does Acid Kill Bacteria?
HCl doubles as a defense barrier. The extreme acidity destroys most pathogens swallowed with food. A 2003 study in Gut journal confirmed that patients on acid-suppressing drugs had significantly higher rates of enteric infections — proving the bactericidal function isn’t optional.
What Happens If There Is Too Much — or Too Little — Acid?
Low HCl (hypochlorhydria) directly impairs protein digestion. Pepsinogen stays inactive, proteins pass partially undigested, and amino acid absorption drops. This also affects B12, iron and zinc uptake, since these nutrients depend on an acidic enviroment for release from food proteins.
A 2013 study in Therapeutic Advances in Drug Safety linked long-term proton pump inhibitor (PPI) use to protein malabsorption and increased fracture risk — likely due to impaired calcium and nutrient processing.
Excess acid, on the other hand, can erode the protective mucin-bicarbonate layer and lead to GERD or peptic ulcers.
Age and HCl: A Factor People Overlook
After age 50, gastric acid output tends to decline. Research published in The American Journal of Clinical Nutrition (1997) showed that up to 30% of older adults have some degree of atrophic gastritis, reducing their ability to digest protein efficiently. This partly explains why elderly individuals often need higher protein intake.
FAQ
What Are the 3 Main Functions of HCl in the Stomach?
Denaturing proteins, activating pepsinogen into pepsin, and killing harmful bacteria.
Does Animal Protein React Differently to HCl Than Plant Protein?
Yes. Animal proteins generally denature faster in acid due to their structure, while some plant proteins (like those in legumes) take longer because of fiber and anti-nutritional factors surrounding them.
How Does Acid Protect the Stomach Lining?
It doesn’t — the mucin layer and bicarbonate ions secreted by surface epithelial cells do. They neutralize H⁺ ions at the mucosal surface, keeping the lining safe while the lumen stays acidic.
Final Thoughts
HCl is the unsung hero of protein digestion. It denatures food proteins, switches on pepsin, guards against infections, and supports micronutrient absorption. When acid levels drop — whether from aging, medication, or disease — protein utilization suffers across the board. Paying attention to your stomach’s acid balance isn’t just about avoiding heartburn; its about making sure every gram of protein you eat actually counts.